Naproxen sodium (NS) is a sodium salt of propionic acid derivative and demonstrates analgesic, anti-pyretic, and anti-inflammatory properties. It exhibits efficiency in being used as an indomethacin in the treatment of acute musculoskeletal disorders. It belongs to the group of nonsteroidal anti-inflammatory drugs (NSAIDS) and acts by inhibiting the activity of both the cyclooxygenase enzymes, COX-1 & COX-2 and further blocking the synthesis of certain prostaglandins. It is administered in the treatment of ankylosing spondylitis, osteoarthritis, rheumatoid disorders, acute gout, mild to moderate pain, tendonitis, bursitis, dysmenorrhea, fever, and migraine headache.
[1][2][3][4][5] Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
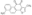 丹参酮I,Tanshinone I;analytical standard, ≥98% (HPLC)
丹参酮I,Tanshinone I;analytical standard, ≥98% (HPLC)![显示明细 6-苄氨基嘌呤,6-Benzylaminopurine [6-BAP, 6-BA];PESTANAL®, analytical standard, ≥99.0% (HPLC) 图片 6-苄氨基嘌呤,6-Benzylaminopurine [6-BAP, 6-BA];PESTANAL®, analytical standard, ≥99.0% (HPLC)](https://www.bszh.cn/images/thumbs/0015330_6-6-benzylaminopurine-6-bap-6-bapestanal-analytical-standard-990-hplc_32.png) 6-苄氨基嘌呤,6-Benzylaminopurine [6-BAP, 6-BA];PESTANAL®, analytical standard, ≥99.0% (HPLC)
6-苄氨基嘌呤,6-Benzylaminopurine [6-BAP, 6-BA];PESTANAL®, analytical standard, ≥99.0% (HPLC)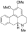 荷叶碱,Nuciferin
荷叶碱,Nuciferin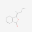 藁本内酯,Ligustilide;≥98%
藁本内酯,Ligustilide;≥98%![显示明细 二氢茉莉酮酸甲酯,Methyl dihydrojasmonate [MDJ];analytical standard, ≥96.0% (GC) 图片 二氢茉莉酮酸甲酯,Methyl dihydrojasmonate [MDJ];analytical standard, ≥96.0% (GC)](https://www.bszh.cn/images/thumbs/0013056_methyl-dihydrojasmonate-mdjanalytical-standard-960-gc_32.jpeg) 二氢茉莉酮酸甲酯,Methyl dihydrojasmonate [MDJ];analytical standard, ≥96.0% (GC)
二氢茉莉酮酸甲酯,Methyl dihydrojasmonate [MDJ];analytical standard, ≥96.0% (GC)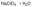 高氯酸钠一水合物,Sodium perchlorate monohydrate;ACS reagent, 85.0-90.0% (monohydrate)
高氯酸钠一水合物,Sodium perchlorate monohydrate;ACS reagent, 85.0-90.0% (monohydrate)
